At the congress of the German Society for Sleep Medicine (DGSM), Julian Mollin was awarded the Children's Sleep Prize 2024 for his outstanding work. He received the award for his published results and the overall project 'Frequency Analysis of Restless Legs Syndrome (RLS) in Children and Adolescents'.
An important part of this project was the development of an algorithm for diagnosing RLS in children and adolescents. This algorithm, based on a sound combination of trial data, clinical experience and relevant literature, provides a structured approach that leads to a consistent and accurate diagnosis. At the same time, it helps to make informed treatment decisions that meet the individual needs of young RLS patients.
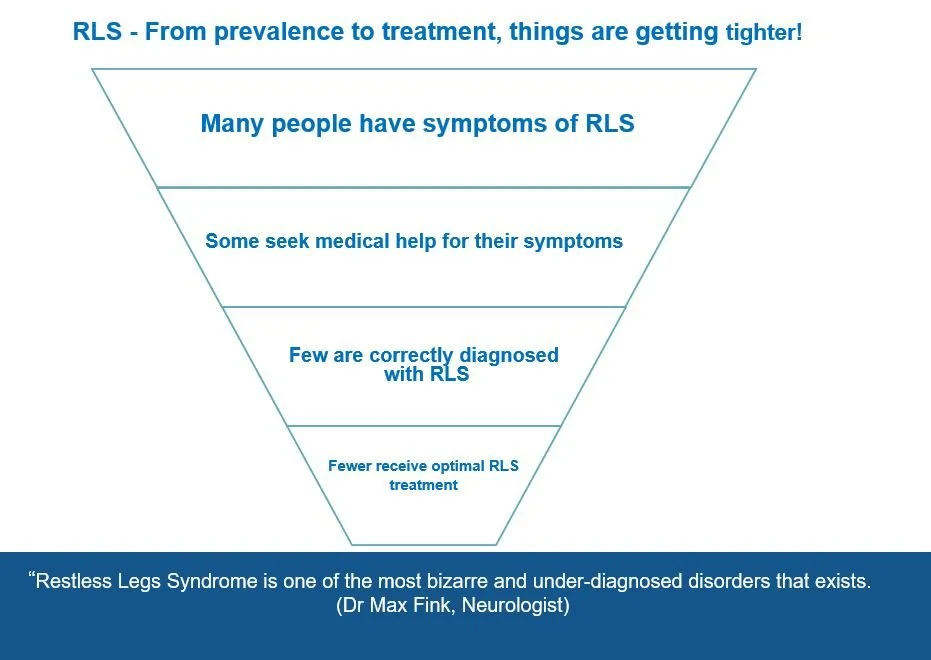
Mollin's research is helping to raise awareness of RLS in children and adolescents and to improve medical care for this patient group.
The study results (in German) can be found here: https://www.springermedizin.de/content/pdfId/27378714/10.1007/s11818-024-00471-6